
Muscle Tissue
TYPES OF MUSCLE TISSUE
Muscle occurs in three distinct types. Click on the thumbnail for a bigger image, or on "More" for more text.
More
More
More
CELLULAR ORGANIZATION OF SKELETAL MUSCLE FIBERS
Unlike most tissues, skeletal muscle does not consist of individual cells. Rather it is formed from huge, multinucleate muscle fibers, which develop by fusion of many individual embryonic cells called myoblasts. Muscle fibers can grow by continued nuclear division within the fibers. Although skeletal muscle fibers are thus not proper, individual cells, the term "muscle cell" is commonly used to refer to one multinucleate fiber. Each individual skeletal muscle fiber extends over much of the length of the muscle in which it resides (up to many centimeters), with a uniform diameter that is typically around 50 µm. In contrast, cardiac muscle (which like skeletal muscle is also striated) and smooth muscle both consist of single, discrete cellular units, each with its own nucleus.
Historically, many microscopic features were described in muscle before they were understood in cells generally. As a result, the distinctive structure of muscle has its own classical terminology. The unique contractile cytoplasm of muscle fibers is called sarcoplasm (sarco = flesh). The contractile machinery is concentrated into myofibrils (myo = muscle), long narrow structures (1-2 µm in diameter) which extend the length of the fiber and form the bulk of the sarcoplasm. Mitochondria (once called sarcosomes) and sarcoplasmic reticulum (a highly specialized form of endoplasmic reticulum) surround each myofibril. The plasma membrane of muscle fibers is sometimes called the sarcolemma.
The many nuclei within each fiber are displaced from the center of the fiber by the specialized sarcoplasm and are located adjacent to the sarcolemma (except in special cases, like the intrafusal fibers of muscle spindles, and regenerated fibers). Any particular cross section of a muscle fiber may reveal one or a few nuclei, or none, but the entire length of the fiber may include hundreds.

Each myofibril is marked by regularly spaced bands (striations) along its length. In relaxed muscle, a single repeat of this pattern is about 2 µm long (the length varies with degree of contraction) and is called a sarcomere. Because the striations on adjacent myofibrils are usually aligned, the entire muscle fiber appears uniformly striated.
- In each sarcomere, the broad band which appears dark in standard histological procedures is called the A-band. This band indicates the location of thick filaments (myosin); it is darkest where thick and thin filaments overlap.
- The broad light band between the dark bands is the I-band. The I-band indicates the location where thin filaments (actin) extend beyond the thick filaments.
- A distinct dark line running down the middle of the I-band is the Z-line, where thin filaments are attached end to end. Where thick filaments are attached end-to-end in the center of the A-band is the M-line.
- When muscle is stretched, an H-band appears along the middle of the A-band, between the free ends of the thin filaments.
From Blue Histology (Copyright Lutz Slomianka 1998-2004)
The Contractile Apparatus of Skeletal Muscle
The spatial relation between the filaments that make up the myofibrils within skeletal muscle fibres is highly regular. This regular organisation of the myofibrils gives rise to the cross-striation, which characterises skeletal and cardiac muscle. Sets of individual "stria" within a myofibril correspond to the smallest contractile units of skeletal muscle, the sarcomeres.
Depending on the distribution and interconnection of myofilaments a number of "bands" and "lines" can be distinguished in the sarcomeres:
(The image should be animated, if you watch patiently.)
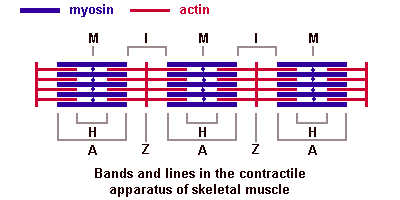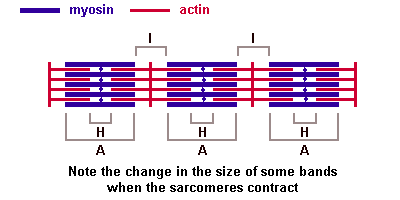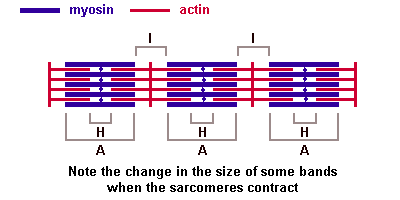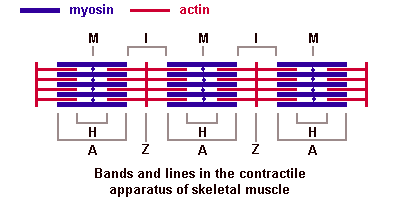
I-band - actin filaments, A-band - myosin filaments which may overlap with actin filaments, H-band - zone of myosin filaments only (no overlap with actin filaments) within the A-band, Z-line - zone of apposition of actin filaments belonging to two neighbouring sarcomeres (mediated by a protein called alpha-actinin), M-line - band of connections between myosin filaments (mediated by proteins, e.g. myomesin, M-protein). The average length of a sarcomere is about 2.5 µm (contracted ~1.5 µm, stretched ~3 µm).
The protein titin extends from the Z-line to the M-line. It is attached to the Z-line and the myosin filaments. Titin has an elastic part which is located between the Z-line and the border between the I- and A-bands. Titin contributes to keeping the filaments of the contractile apparatus in alignment and to the passive stretch resistance of muscle fibres. Other cytoskeletal proteins interconnect the Z-lines of neighbouring myofibrils. Cytoskeletal proteins also connect the Z-lines of the peripheral myofibrils to the sarcolemma.
(These features are nicely displayed in electron micrographs from Universität Mainz.)
Of historical interest: "A" stands for "anisotropic" and "I" for "isotropic," referring to the optical properties of living muscle as demonstrated with polarized-light microscopy. "Z" stands for "zwischen" (German for "between"), "H" for "helle" ("bright"), and "M" for "mittel" ("middle").
Beside each myofibril are numerous mitochondria and regularly spaced sets of three membrane-bound chambers. Each such triad is located adjacent to the boundary between an A-band and an I-band. The middle one of each triad is a transverse tubule, or T-tubule. The transverse tubules are understood to be infoldings of the plasma membrane. On either side of the T-tubule is a terminal cisterna of the sarcoplasmic reticulum, a set of internal membrane-bound spaces which sequester calcium ions. These membranes are involved in "excitation-contraction coupling."
Individual skeletal muscle fibers may differ from one another in several ways, including how quickly they can contract, how readily they will fatigue, and how they metabolize energy. Muscle fibers may be classified as red or white, slow twitch or fast twitch, oxidative or glycolytic. (Although different contrasting pairs are related, the sets are not congruent.) The relative proportions of the fiber types in a muscle influences its gross behavior.
Feel the difference: To experience the difference between fast twitch ("white" muscle, not so much myoglobin) and slow twitch ("red" muscle, lots of myoglobin), try the following. First, blink your eyelid. Now "blink" your anal sphincter. That's the difference between "fast" and "slow".
INNERVATION OF SKELETAL MUSCLE

Each skeletal muscle fiber is innervated by a single motor axon. The same axon may also innervate other muscle fibers. All the fibers innervated by the same axon are called a motor unit. In muscles with delicate action, such as like those moving fingers or eyeballs, the motor units are small, with only one or few fibers; motor units are much larger in muscles with cruder action, like those in the back or thigh.
Muscle action begins at the motor end plate (or neuromuscular junction), which is analogous to a synapse. The neuromuscular junction is a site located midway along the length of a muscle fiber where a motor axon terminal releases transmitter (acetylcholine) into the junctional cleft between axon and muscle membranes. Acetylcholine binds to receptors localized in the muscle membrane at the motor end plate. Resulting local depolarization at the end plate (the "end plate potential") normally exceeds threshold and triggers a depolarizing action potential, just like that which travels along axon membranes. This muscle action potential zips toward both ends of the muscle fiber, accomplishing coordinated activation of the entire length of the fiber by a single axon terminal. (The transverse tubule system conveys this activation deep into the fiber; more below.)
Muscle proprioception -- muscle spindles and stretch reflexes
Skeletal muscles contain specialized proprioceptive sense organs, called muscle spindles, which function to detect muscle stretch.
Each muscle spindle consists of an encapsulated cluster of small striated muscle fibers ("intrafusal muscle fibers") with somewhat unusual structure (e.g., nuclei may be concentrated in a cluster near the middle of the fiber's length). Each fiber has a mechanosensory nerve ending (the most prominent of these are called "annulospiral endings") which wraps around the mid-region of the fiber. This sensor produces nerve impulses in response to stretch. Each fiber also receives motor innervation from a γ (gamma) efferent nerve fiber. Impulses in this fiber cause the spindle muscle fiber to contract.
Stretch reflexes: If a muscle spindle is stretched, sensory impulses ascend Ia afferents to the spinal cord where they monosynaptically trigger motor impulses to the entire muscle; this stimulates contraction to oppose the stretch. Interneurons mediate further reflexes which relax antagonist muscles and inform other muscles to maintain postural balance.
Clinical note: Such reflex action can be tested or demonstrated by delivering a sudden, brief tap to a tendon (e.g., the patellar tendon for the familiar "knee-jerk" response).Gamma efferent (motor) innervation of the muscle spindle permits the length of the spindle muscle fibers to be set to match the overall muscle length. The spindle can thus be used to establish a set-point for muscle length.
Tendon organ drawn by Camillo Golgi, 1903. Additional proprioceptor endings (Golgi tendon organs, named after Camillo Golgi, b. 1843) are located at the point where muscle fibers attach to tendon. These sensitively respond to tension (force) exerted by the muscle; activity in these axons inhibits muscle contraction. And yet other proprioceptors are found in joints. The functions of tendon and joint proprioceptors are less-well understood than those of muscle spindles.
EXCITATION-CONTRACTION COUPLING
In terms of ordinary cell size, a muscle fiber is huge. Yet for coordinated action, the entire fiber must contract together, as a single unit. Synchronization of contraction along the length of the fiber is accomplished by the muscle fiber action potential which zips rapidly along the fiber after being triggered by neurotransmitter release at the motor end plate, upon arrival of a motor axon action potential. Synchronization across the thickness of the fiber is accomplished by conduction of the muscle action potential down the transverse tubules, which are invaginations of surface plasma membrane. (More, at the UCSD Muscle Physiology page.)
The processes which link a depolarization of the muscle fiber surface membrane to contraction of the myofibrils in its sarcoplasm are called excitation-contraction coupling. The membrane of the transverse-tubule lies immediately adjacent to that of the terminal cisternae of the sarcoplasmic reticulum, so ion movement across the former could presumably affect the latter. But the actual mechanism whereby depolarization of the T-tubule membrane causes Ca2+ release by the sarcoplasmic reticulum remains obscure. Once released into cytoplasm, Ca2+ diffuses into the myofibrils where it binds to troponin and triggers several protein conformational changes which together cause forcible sliding of thick filaments along thin filaments. Active calcium reuptake by the sarcoplasmic reticulum ends the contraction. (More, at the UCSD Muscle Physiology page.)
ASSOCIATED TISSUES
Each skeletal muscle fiber is invested by a delicate support of connective tissue called endomysium. Within the endomysium is an extensive network of capillaries around each fiber to provide oxygen and nutrition. Also found adjacent to muscle fibers are occasional satellite cells, inconspicuous stem cells which may divide and differentiate to regenerate muscle after injury. Bundles of muscle fibers within a muscle are separated by connective tissue called perimysium, while the entire muscle is ensheathed by connective tissue call epimysium.
When you examine skeletal muscle through the microscope, nuclei of fibroblasts, capillary endothelium and satellite cells may all be seen within the thin endomysium, adjacent to the nuclei of the muscle fibers. These various nuclei can be difficult to distinguish from one another in routine preparations.
The endomysium permits the forces of contraction to be passed from each muscle fiber toward the ends of the muscle where the fibrocollagenous tissue of endomysium, perimysium, and epimysium merge to form tendon, fascia, or direct attachment to bone.
SKELETAL MUSCLE PATHOLOGY
Washington University Neuromuscular Disease Center hosts an excellent web resource for neuromuscular pathology.
Within this site, you can find Differential Diagnosis for myopathy and neuropathy, and much more.Muscle fiber atrophy in ALS (amyotrophic lateral sclerosis) is illustrated at Webpath.
Comments and questions: dgking@siu.edu
SIUC / School
of Medicine / Anatomy / David
King
https://histology.siu.edu/ssb/muscle.htm
Last updated: 1 January 2023 / dgk